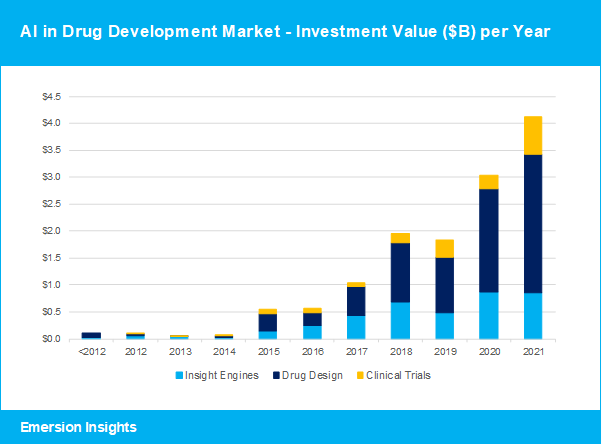
Funding for artificial intelligence in drug development hit $4.1B in 2021, a 36% increase from the year before. The AI drug development industry has gained a lot of attention from investors, venture capital firms and corporate investment funds in recent years, now culminating in a total investment in the industry exceeding $13B and many pharma partnerships ensuring promising revenues in the coming years.
AI drug designers continue to receive the largest proportion of funding in the AI drug development industry. These applications are using AI to develop new molecules, predict and optimise drug specificity and efficacy, or to select drug repurposing options for existing drugs. Some of the AI drug designers who received funding in 2021 include XtalPi, Insitro, Exscientia and Insilico Medicine.
Insight engines are artificial intelligence platforms that aggregate and analyse information and real-world evidence from multiple sources such as scientific literature, patient data and clinical trial information to find new associations and guide drug discovery or clinical trial optimisation. Some of the insight engine vendors funded in 2021 include PatSnap, iCarbonX, Paige, Cellarity and Aetion.
Clinical trial applications help optimise the clinical trial process by improving patient stratification with finer nuances to get the right patients for the right trials, optimise enrolment and retention, and in some cases help reduce the number of patients needed for a successful trial. Some of the clinical trial vendors funded in 2021 include Medable, PathAI and Athelas.
Although initial funding rounds still exist in the industry, investors are increasingly doubling down on their existing portfolio companies. Already successful and well-funded vendors receive larger proportions of the new funding, indicating an increasing consolidation of investments in the industry. The average funding size jumped from $51 million in 2020 to $79 million in 2021, an increase driven by some very large Series C and D funding rounds.
Clinical trial AI solutions received more funding in 2021. One consequence of the COVID-19 pandemic is that clinical trial speed and efficiency have become favourite topics around the dinner table. The importance of optimising this crucial step in the drug development process is now widely understood. And as the technologies to support these processes have gained strength, so has the financial support from healthtech investors. The annual funding for clinical trial AI technologies jumped from $255 million in 2020 to $704 million in 2021, while the average funding size in this category increased to $64 million.
Over the next 12 months, we will continue to see massive funding rounds in this industry as it matures. A large proportion of these investments will come from corporate investment funds placing strategic investments in their AI partners. A few large pharmaceutical deals are expected to reach clinical milestones this year, which will result in sizeable milestone payments to AI specialists. Vendors reinvesting these funds in creating multi-therapeutic platforms, corresponding to their pharmaceutical partners product portfolios, will be able to strengthen their competitive position and build on existing partnerships in the pharmaceutical industry.
More Research Available
Emersion Insights specialise in strategic market intelligence for investors and entrepreneurs. Our AI in Drug Development market report includes detailed investment analysis, market sizing, forecasting and strategic insights to help entrepreneurs and investors navigate in this fast-moving industry. Reach out to Founder & Principal Analyst Dr Ulrik Kristensen for more information: hello@emersioninsights.io